Choosing a Luciferase Reporter Assay
A reporter assay comparison guide.
Luciferase Reporters and Detection Reagents
Since developing the first commercially available luciferase assay reagents in 1991, our scientists have continued engineering bioluminescent reporter technologies, from optimized firefly luciferase through NanoLuc® luciferase, a 19 kDa enzyme approximately 100-fold brighter than conventional reporters. Today this work spans genetic reporter assays, protein interaction studies, target engagement and live-cell imaging. This guide will help you choose the right luciferase reporter system for your experimental goals.
Key Takeaways
- Three distinct luciferase reporters (NanoLuc, firefly, Renilla) are each engineered with different size, brightness and substrate properties to match specific experimental designs.
- NanoLuc® luciferase is approximately 100-fold brighter than firefly or Renilla at equivalent expression levels when maximum sensitivity is needed for low-expression targets or limited samples.
- Homogeneous "add-and-read" reagents (Nano-Glo, Bright-Glo, ONE-Glo) eliminate separate lysis steps and are designed for high-throughput, plate-based workflows.
- Dual-reporter formats, pairing a firefly experimental reporter with a Renilla or NanoLuc® normalization control, are the standard approach for transcriptional studies where transfection efficiency must be controlled.
- Live-cell substrates (Endurazine, Vivazine, EnduRen, ViviRen) enable non-lytic, kinetic measurements from the same cell population over hours to days for time-course and longitudinal experiments.
Which luciferase reporter is right for my experiment?
When creating a luciferase reporter assay, there are two important elements to consider: the luciferase reporter protein itself and the assay chemistry used to detect reporter activity. The characteristics of these two components together contribute to the overall performance of the assay. By selecting the reporter/detection solution that is optimal for your experimental goals, you can customize your luciferase reporter assay to create the best solution for your research.
Luciferase Reporter Comparison
Promega offers a choice of 3 different luciferase reporters: NanoLuc® Luciferase, Renilla Luciferase and Firefly Luciferase. See table below to compare their key properties. Learn more in this article: Reporter Genes and Their Applications.
| Luciferase Reporter | |||||
|---|---|---|---|---|---|
| Firefly Luciferase | |||||
| NanoLuc® Luciferase | |||||
| Renilla Luciferase |
*Destabilized versions available to more tightly couple to transcriptional response.
**Secreted versions available.
Detection Reagent Considerations
Multiple assay detection reagents are also available for each reporter. Key considerations for selecting the optimal assay reagent include:
- Signal intensity and overall dynamic range needed for the assay.
- Signal stability, or half-life, which will impact your processing workflow.
- Processing steps required. Non-homogenous assays require a separate lysate creation step prior to reagent addition. Homogenous assay reagents are added directly to the cells in culture eliminating sample pre-processing.
- Lytic or live-cell reporter detection
- Single or dual-reporter detection
How do signal strength and stability differ across luciferase assay systems?
How do I choose a luciferase assay system?
| Luciferase Detected | Assay Reagent | Ideal For | Signal Half-Life | Sensitivity | Number of Steps | Live Cell Assay? | Injectors Needed? |
|---|---|---|---|---|---|---|---|
| HiBiT, Firefly | Nano-Glo® HiBiT Dual-Luciferase® Assay System (HiBiT NanoDLR™) | Homogenous Fluc/HiBiT dual-reporter detection. Provides high sensitivity for both direct protein quantification via HiBiT tagging and an internal control or downstream transcriptional readout via firefly. Compatible with low to high-throughput processing. | 2 hours each | ++++ (HiBiT) ++ (Fluc) |
2 (lysate optional) | No | No |
| NanoLuc, Firefly | Nano-Glo® Dual-Luciferase® Reporter Assay System (NanoDLR) | Homogenous Fluc/Nluc dual-reporter detection with flexibility in choice of primary reporter. Provides highest sensitivity for both reporters when a stable signal is required and highest sensitivity option when Nluc is used as the primary reporter. Ideal for low to high-throughput processing when 2 primary reporters or internal control is needed. | 2 hours each | +++++ (Nluc) ++ (Fluc) |
2 (lysate optional) | No | Optional |
| Firefly, Renilla |
Dual-Luciferase® Reporter Assay System (DLR) | Non-homogenous Fluc/Rluc dual-reporter detection. Requires lysate creation and 2 injectors for delivery. Ideal for small sample numbers when maximum Fluc sensitivity and internal control are needed. | 10 minutes, 2 minutes | ++++ (Fluc) ++++ (Rluc) |
3 | No | Yes 2 injectors |
| Firefly, Renilla |
Dual-Glo® Luciferase Assay System | Homogenous Fluc/Rluc dual-reporter detection with reduced sensitivity and stable signal. Ideal for high-throughput processing of Fluc reporter when Rluc is used as the internal control. | 2 hours each | + (Fluc/Rluc) |
2 | No | No |
| NanoLuc | Nano-Glo® Luciferase Assay System | Homogenous Nluc detection with bright, stable signal. Ideal for low to high-throughput processing when maximum sensitivity is required. | 2 hours | +++++ | 1 | No, unless used with secNluc | No |
| NanoLuc | Nano-Glo® Live Cell Assay System | Live cell Nluc detection with brightest signal. Ideal for single timepoint analysis when high sensitivity is needed in a non-lytic assay. | Up to 2 hours | ++++ | 1 | Yes | No |
| NanoLuc | Nano-Glo® Vivazine™ Live Cell Substrate | Live cell Nluc detection with intermediate signal and stability. Ideal for kinetic analysis lasting multiple hours. | Up to 24 hours | +++ | 1 | Yes | No |
| NanoLuc | Nano-Glo® Endurazine™ Live Cell Substrate | Live cell Nluc detection with most stable signal. Ideal for multi-day kinetic analysis. | Up to 72 hours | ++ | 1 | Yes | No |
| Firefly | Luciferase Assay System | Non-homogenous Fluc detection requiring lysate creation and injector delivery. Ideal for small sample numbers when maximum Fluc sensitivity is needed. | 10 minutes | ++++ | 2 | No | Yes |
| Firefly | Bright-Glo® Luciferase Assay System | Homogenous Fluc detection offering the brightest signal and shortest half-life. Ideal for high-throughput processing when high sensitivity is required. | 30 minutes | +++ | 1 | No | No |
| Firefly | ONE-Glo™ Luciferase Assay System | Homogenous Fluc detection with moderate signal and half-life. Ideal for high- or ultrahigh-throughput processing. | 45 minutes | ++ | 1 | No | No |
| Firefly | ONE-Glo™ Ex Luciferase Assay System | Homogenous Fluc detection with moderate signal and half-life and improved storage stability. Ideal for high- or ultrahigh-throughput processing and repeat use. Also used to detect Fluc in NanoDLR allowing consistent use between single and dual assays. | 2 hours | ++ | 1 | No | No |
| Firefly | Steady-Glo® Luciferase Assay System | Homogenous Fluc detection with maximum signal stability and reduced signal. Ideal for high-throughput applications when extended luminescence is required. | 5 hours | + | 1 | No | No |
| Renilla | Renilla Luciferase Assay System | Non-homogenous Rluc detection requiring lysate creation and injector delivery. Ideal for small sample numbers when maximum Rluc sensitivity is needed. | 2 minutes | ++++ | 2 | No | Yes |
| Renilla | Renilla-Glo Luciferase Assay System | Homogenous Rluc detection with increased signal stability. Ideal for high-throughput processing. | 40+ minutes | ++ | 1 | No | No |
| Renilla | ViviRen™ Live Cell Substrate | Live cell Rluc detection with highest signal. Ideal for single time point or short non-lytic analysis when highest sensitivity is required. | 10 minutes | +++ | 1 | Yes | No |
| Renilla | EnduRen™ Live Cell Substrate | Live cell Rluc detection with greatest stability. Ideal for extended kinetic analysis. | Up to 24 hours | + | 1 | Yes | No |
What should I consider when designing a bioluminescent reporter assay?
Interested in learning more about reporter assay design? Our two-part "Designing a Bioluminescent Reporter Assay" guide will walk you through basic considerations for choosing the optimal experimental reporter, experimental design and data analysis methods.
Related Resources
Guide: Reporter Genes and their Applications

Webinar: Understanding Reporter Assay Design
Blog: Tips for Performing Dual-Reporter Assays
Frequently Asked Questions About Luciferase Reporter Assays
What is the difference between NanoLuc® and firefly luciferase?
NanoLuc® is a 19 kDa enzyme engineered by our scientists from a deep-sea shrimp luciferase. It produces approximately 100-fold brighter signal than firefly luciferase (61 kDa) using the substrate furimazine. Its small size makes it a better fusion partner for studying protein dynamics without disrupting target protein function, while firefly luciferase remains widely used for transcriptional reporter assays and is well suited for dual-reporter experiments paired with Renilla luciferase.
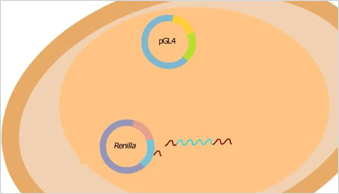
When should I use a dual-reporter luciferase assay?
Dual-reporter assays are recommended whenever you need an internal control for experimental variability — particularly transfection efficiency. The standard approach pairs a firefly luciferase experimental reporter with a Renilla or NanoLuc® control reporter, measured sequentially from the same sample. Our Dual-Luciferase® and NanoDLR™ assay systems are designed for this workflow.
Can I measure luciferase activity in live cells without lysis?
Yes. Our live-cell substrates (Endurazine and Vivazine for NanoLuc, EnduRen and ViviRen for Renilla) are cell-permeable prodrugs that are converted to active substrate inside living cells. This enables real-time, kinetic measurements over hours to days from the same cell population, which is essential for time-course studies and longitudinal experiments.
What luciferase assay format works best for high-throughput screening?
Homogeneous, single-addition reagents are designed for HTS workflows because they eliminate wash and lysis steps. Bright-Glo™ and ONE-Glo™ systems (firefly) and Nano-Glo (NanoLuc) are add-and-read formats compatible with 384- and 1536-well plates. For dual-reporter HTS, the Nano-Glo® Dual-Luciferase® Reporter (NanoDLR™) system provides the best combination of sensitivity and throughput.
How do I choose between flash and glow luciferase assay formats?
Flash assays (e.g., Luciferase Assay System) produce an intense but short-lived signal, ideal for single-sample or tube-format measurements with injector-equipped luminometers. Glow assays (e.g., Bright-Glo, ONE-Glo, Nano-Glo) produce a stable signal lasting minutes to hours, which is necessary for batch processing and plate-based reading. For most plate-based experiments, glow-format reagents are the practical choice.
Beyond Reporter Assays: Our Bioluminescence Platform
The luciferase reporter you choose today connects you to a broader ecosystem of bioluminescence technologies we've developed. NanoLuc® luciferase is the foundation for NanoBRET™ target engagement assays, NanoBiT® protein interaction systems, HiBiT protein tagging, and Lumit® immunoassays. We've designed them all to work together so you can follow your biology from initial reporter experiments through compound screening, protein dynamics and live-cell imaging with the GloMax® Galaxy Bioluminescence Imager. One integrated platform represents more than 30 years of continuous innovation.

